Why the Plant’s Native Chemistry Has Been Systematically Underinvestigated
Chris Emerson, PhD Founder, Chief Scientist, and CEO, LEVEL
Metta Medical Inc dba LEVEL
Chris Emerson holds a doctorate in small-molecule chemistry from Oregon State University. He is the founder, chief scientist, and CEO of Metta Medical dba LEVEL. His publications appear in Organic Letters, Chemistry: A European Journal and Chemical Communications. He holds US Patents 12,076,442 B1 and 11,596,606 B2 for activated cannabinoid controlled-release formulation technology. He is an employee, shareholder, and fiduciary officer of Metta Medical dba LEVEL.
SECTION I
Acidic Cannabinoids as Primary Biosynthetic Output: Correcting the Transformation Bias in Cannabis Science
The cannabis plant does not biosynthesize THC or CBD. It biosynthesizes their acidic precursors: cannabigerolic acid (CBGa), tetrahydrocannabinolic acid (THCa), and cannabidiolic acid (CBDa). These acidic cannabinoids are the plant’s primary biosynthetic chemical output. THC and CBD are formed when heat, time, oxidation, or photochemical exposure removes the carboxyl group in a process called decarboxylation.
Modern cannabinoid science has extensively characterized THC and CBD, the molecules most abundant after heat-driven transformation. The acidic forms most abundantly produced by the living plant remain comparatively underexplored. This imbalance is not a reflection of biological relevance. It is a structural artifact of how the field developed: shaped by combustion-based consumption, early analytical limitations, and regulatory constraints that narrowed inquiry toward the psychoactive properties of cannabinoids.
This guide proceeds from a governing thesis: acidic cannabinoids, specifically CBGa, THCa, and CBDa, represent a structurally and pharmacologically distinct molecular class whose peripheral distribution bias, non-CB1 receptor engagement, and preserved biological activity after oral dosing are not reducible to in vivo decarboxylation and warrant scientific investigation commensurate with their thermally transformed neutral counterparts. That thesis is evaluated through structural chemistry, stability science, physiological distribution logic, lipid signaling cross-talk, and observational clinical data from a 12-week study conducted with investigators affiliated with the University of Michigan involving 162 participants with fibromyalgia, rheumatoid arthritis, and osteoarthritis.
The purpose of this document is not to argue for the superiority of acidic cannabinoids over their neutral counterparts. Rather, it is to ask whether the plant’s primary biosynthetic output has received proportional scientific attention and to present the current evidence for why that question matters.
What Does the Plant Actually Biosynthesize?
Modern cannabis science has disproportionately studied the plant’s transformed chemical entities rather than those present in its native biosynthetic state. This imbalance is not accidental. Early scientific inquiry into cannabis was shaped by legal restriction, cultural focus on intoxication, and the practical realities of combustion-based exposure. As analytical chemistry matured, research gravitated toward the molecules most responsible for psychoactivity and most abundant after heat exposure.
For most of recorded history, human interaction with cannabis has involved heat. Combustion, vaporization, cooking, and prolonged drying introduce energy into the plant matrix. Heat drives decarboxylation. Time, oxidation, and light further reshape molecular structure.[1,2,3]
The cannabinoids most commonly studied, THC and CBD, are not the compounds the plant natively synthesizes in living tissue. They are transformation products.
The cannabis plant does not biosynthesize CBG, CBD, or THC directly. It biosynthesizes their upstream acidic precursors: CBGa, CBDa, and THCa. Acidic cannabinoids are the plant’s primary biosynthetic chemical output.[4,5]
How Does Biosynthesis Proceed Before Transformation?
Within the living plant, cannabinoids exist predominantly in their carboxylated form. Cannabigerolic acid (CBGa) serves as the biochemical precursor from which THCa, CBDa, and CBCa are enzymatically formed through dedicated synthase enzymes.[4,5]
The carboxyl group, a CO2-containing moiety, is not incidental. It is the direct product of enzymatic biosynthesis within glandular trichomes. Only after senescence, harvest, or environmental exposure does meaningful molecular transformation begin.
Decarboxylation, or the loss of CO2, occurs through several mechanistic pathways, including thermal energy input, oxidative exposure, photochemical degradation, surface-area effects due to mechanical disruption, and time-dependent spontaneous conversion.
Why Did Early Research Focus on Transformed Rather Than Native Chemistry?
Early cannabinoid research emerged alongside interest in psychoactivity and as chemical tools became robust enough to isolate, characterize, and synthesize these compounds. THC, the abbreviated reference to delta-9 tetrahydrocannabinol, the decarboxylated form of THCa, binds directly to CB1 receptors in the brain and produces intoxicating effects.[6] Combustion-based consumption ensured that most exposures involved decarboxylated cannabinoids.
Scientific investigation followed the most visible and measurable effects, and legal restrictions further narrowed what was studied and how. This convergence of psychoactivity, combustion, and regulatory constraint created a structural bias. The compounds most thoroughly studied, however, are not the molecules most abundantly produced by the living plant. Rather, they are the molecules most abundantly produced after environmental transformation.
Is Decarboxylation the Same as Activation?
Decarboxylation is often described as activation, as though removing a functional group represents biological improvement. Chemically, it is a structural transformation that changes distribution, receptor interaction, and membrane permeability.
Structurally, removal of the carboxyl group alters polarity, lipophilicity, hydrogen-bonding capacity, membrane permeability, and conformational geometry. These chemical changes influence both pharmacokinetic and pharmacodynamic interactions between the molecules and human physiology.[7]
Acidic cannabinoids and their neutral derivatives are not, respectively, stronger and weaker versions of the same compound. They are structurally distinct molecules with distinct pharmacokinetic and pharmacodynamic profiles.
How Does the Historical Record of Human Exposure Inform This Question?
Cannabis has been cultivated for at least 5,000 years of recorded human history.[8,9] Human exposure likely included both raw and partially transformed preparations. The exact proportions of acidic versus neutral exposure are unknown. What is clear is that acidic cannabinoids are not modern synthetic artifacts. They are intrinsic to the plant’s longstanding biochemical identity.
Modern analytical chemistry now enables the preservation and quantification of acidic cannabinoids without inducing artificial decarboxylation. This creates an opportunity to revisit questions that early combustion-dominated research could not adequately address.[10]
What Is the Governing Thesis of This Guide?
Every scientific field is shaped by the tools and assumptions available at the time. Early cannabinoid science was shaped by historical precedent, the development of analytical chemistry, and regulatory constraints.
Today, we have the analytical capacity to investigate and understand the plant’s native state without first altering it. If structure determines interaction, and interaction determines effect, then preserving molecular topology is not merely a manufacturing preference. It is a commitment to biological fidelity.
Modern cannabinoid research has extensively mapped the pharmacology of chemically transformed cannabinoids. The plant’s primary biosynthetic output remains comparatively underexplored.
The question is not whether acidic cannabinoids are better but whether we have studied the molecules the plant natively biosynthesizes with the same rigor applied to those created through chemical transformation.
The following sections examine that thesis through structural chemistry, physiological distribution, lipid signaling cross-talk, stability science, and real-world observational data from 162 participants. Section V presents that observational evidence, including large-effect-size outcomes in pain interference, sleep disturbance, and anxiety across chronic pain populations.
SECTION II
CBGa, Carboxyl Group Chemistry, and Why Decarboxylation Is Structural Transformation, Not Activation
To understand acidic cannabinoids, we begin where the plant begins: with CBGa.
What is CBGa, and Why Is it the Molecular Origin Point of Cannabinoid Biosynthesis?
Cannabigerolic acid (CBGa) is the central precursor in cannabinoid biosynthesis. Within glandular trichomes, specialized oxidocyclase synthase enzymes convert CBGa into THCa, CBDa, and CBCa via their respective synthases.[4,5]
These transformations are enzymatically regulated within the plant’s secondary metabolite system. The carboxyl group is not incidental; it is intrinsic to the biosynthetic architecture these enzymes produce. In living tissue, cannabinoids are overwhelmingly present in their carboxylated state.
How Does the Carboxyl Group Function as a Structural Modifier Rather Than an Inert Appendage?
The difference between THCa and THC is the presence of a single carboxyl group (-COOH). Removal of this group as CO2 produces the neutral molecule. This structural modification alters several fundamental physicochemical properties, including increased lipophilicity following decarboxylation, reduced polarity, altered hydrogen-bonding potential, and changes in molecular geometry that influence membrane diffusion behavior.[7,11]
Small structural changes can produce meaningful pharmacokinetic consequences. Structure–activity relationships (SAR) dictate that distribution patterns and receptor interaction probabilities shift with changes in topology. Acidic and neutral cannabinoids are structurally related but pharmacologically distinct molecules.[7]
If acidic cannabinoids are the plant’s primary biosynthetic chemical output, then structural independence from their neutral derivatives is not a minor detail but a foundational distinction.[11]
Through Which Pathways Does Decarboxylation Occur, and What are Their Relative Contributions?
Decarboxylation is often described as a heat-driven process. That description is accurate but incomplete.
Transformation of acidic cannabinoids can occur via several pathways, including thermal decarboxylation (combustion, vaporization, baking), oxidative degradation, photochemical exposure, time-dependent spontaneous conversion, and surface-area-dependent acceleration.[1,2]
Thermal input dramatically accelerates CO2 loss. However, environmental exposure alone can gradually shift acidic cannabinoid topology, even at room temperature. Neutral cannabinoids are environmentally transformed derivatives of the plant’s natural biosynthetic output.[1]
How Does the Carboxyl Group Influence Physiological Distribution?
The presence or absence of the carboxyl group meaningfully influences distribution within human physiology.
Neutral cannabinoids such as THC are highly lipophilic and readily cross the blood-brain barrier (BBB), enabling central CB1 receptor activation and psychoactive effects.[12]
Acidic cannabinoids, by contrast, are more polar. Increased polarity generally reduces passive diffusion across lipid membranes.[11,13] The brain-to-plasma ratio for phytocannabinoid acids has been measured at 0.04 or below, consistent with markedly reduced central nervous system penetration relative to their neutral counterparts.[13]
Reduced central nervous system penetration is consistent with lower CB1 receptor engagement, absence of intoxicating psychoactivity, and greater relative peripheral signaling. These distributional differences are structural consequences. Pharmacodynamics is influenced by topology as much as by receptor affinity.[12,13]
What Does the Threshold of Toxicological Concern Framework Offer, and Where Does It Fall Short?
Acidic cannabinoids are less extensively studied than their neutral counterparts in peer-reviewed clinical research. However, structurally, they share core cannabinoid scaffolds with well-characterized compounds.[14,15]
The Threshold of Toxicological Concern (TTC) framework was developed to estimate acceptable exposure limits for trace-level contaminants in food matrices, based on structural class and known toxicophore patterns.[14,15] Strictly applied, TTC is not designed for intentionally dosed bioactive compounds at therapeutic concentrations, and invoking it as a primary safety basis for acidic cannabinoids would misrepresent its scope.
Nevertheless, the framework offers a rational preliminary lens for a specific, limited purpose: establishing that acidic cannabinoids do not introduce novel structural toxicophores outside the broader cannabinoid chemical class. Given that cannabinoids as a class have an extensive human exposure history and an established safety reference set, the structural relatedness of THCa and CBDa to well-characterized compounds provides a rational foundation for cautious scientific exploration rather than categorical dismissal. This does not eliminate the need for dedicated, long-term human safety data specifically for acidic cannabinoids. Such data do not yet exist in sufficient volume, and that gap is acknowledged directly.[14,15]
What is the Current State of Evidence on In Vivo Decarboxylation, and Why Does This Assumption Warrant Reexamination?
For decades, it has been hypothesized that acidic cannabinoids rapidly decarboxylate upon ingestion, effectively converting into their neutral forms in vivo.[16]
If complete and immediate decarboxylation occurred systemically, one would expect no measurable acidic cannabinoids in circulation, as well as no distinct pharmacological signals attributable to acidic forms. In essence, the effects would be indistinguishable from neutral analogues.
However, limited pharmacokinetic data suggest that acidic cannabinoids can be detected in plasma following oral ingestion.[17] No specific human decarboxylase enzyme has been identified that catalyzes the rapid conversion of cannabinoid acids in vivo.[13,17] Low gastric pH alone does not guarantee spontaneous CO2 loss. Carboxylic acids do not uniformly decarboxylate under physiological temperature and aqueous conditions without catalytic or energetic assistance.
Empirical observation suggests that acidic cannabinoids produce effects distinct from their neutral counterparts, and emerging peer-reviewed evidence indicates that in vivo conversion may be partial rather than complete.[17] Therefore, the longstanding assumption of rapid, total decarboxylation in the body remains unsettled and warrants careful re-examination.
SECTION III
Peripheral Distribution, COX Modulation, and Endocannabinoid System Cross-Talk: The Physiological Case for Acidic Cannabinoids
Whereas Section II outlines the structural differences between acidic and neutral cannabinoids, Section III examines the physiological implications of those differences. The distinction is not whether acidic cannabinoids are active; it is where and how they may interact physiologically. Central psychoactivity is readily perceived and readily measured; peripheral modulation is not. This difference in perceptibility has influenced which molecular effects receive the most scientific attention.
How Does Reduced Central Penetration Shift the Site and Character of Potential Pharmacological Interaction?
THC exerts psychoactivity primarily by activating central CB1 receptors.[6] Acidic cannabinoids display weak direct CB1 agonism compared with THC.[18] Given their increased polarity and expected lower blood-brain barrier penetration,[13] acidic cannabinoids may demonstrate relatively greater peripheral engagement.
Peripheral tissues contain both classical and nonclassical cannabinoid-responsive pathways, including CB2 receptors, TRP channels, various enzyme targets, and immune-modulating signaling cascades.[19,20] Modulation without central dominance may produce subtle systemic effects without intoxication. Subtle does not mean insignificant. It may reflect distributed signaling rather than concentrated central activation. Empirically observed subtlety is consistent with expected distribution patterns and receptor interaction profiles.
What Does CBDa’s COX Enzyme Interaction Establish About Mechanism, and Where Does Translational Uncertainty Persist?
CBDa has demonstrated in vitro inhibition of COX-1 and COX-2 enzymes.[21] These enzymes convert arachidonic acid into prostaglandins, key mediators of inflammatory signaling. Nonsteroidal anti-inflammatory drugs target this same enzymatic pathway.
In vitro inhibition does not automatically translate into clinical anti-inflammatory efficacy. However, it establishes mechanistic plausibility that is distinct from receptor-driven psychoactivity. Much of cannabinoid pharmacology has historically been interpreted through receptor binding models. Enzyme interaction has received comparatively less conceptual emphasis. Structural differences between acidic and neutral cannabinoids may influence enzyme binding geometry, an active area of research.
One important qualification applies to interpreting this finding. In vitro enzyme inhibition studies establish mechanistic plausibility but do not confirm clinical efficacy. The concentrations of CBDa required to produce COX-2 inhibition in the Takeda et al. experimental system must be considered alongside the plasma concentrations achievable through oral dosing in humans.[21] If therapeutically relevant CBDa plasma levels fall significantly below the concentrations demonstrated to inhibit COX-2 in vitro, the in vitro finding loses direct translational weight, even while retaining mechanistic interest. Whether oral CBDa dosing in humans produces plasma concentrations sufficient for COX inhibition has not been confirmed in published human pharmacokinetic trials. This remains an open and clinically important question, and resolving it represents a high-value target for future research.
What Does CBDa’s 5-HT1A Agonism Indicate About Structural Independence From CBD?
Among the most pharmacologically significant findings in acidic cannabinoid research is the demonstration by Bolognini and colleagues (2013) that CBDa acts as a potent and selective agonist at the 5-HT1A receptor, with greater potency at this receptor than CBD itself.[18] The 5-HT1A receptor is a well-characterized target in the pharmacology of nausea, anxiety, pain modulation, and stress response. It is the primary target of buspirone and a secondary target of several antidepressant agents.
This finding is significant for several reasons. First, it provides a mechanistic pathway for CBDa to exert peripheral and central modulatory effects that are entirely independent of CB1 receptor engagement and therefore structurally consistent with non-intoxicating biological activity.[18]
Second, the observation that CBDa demonstrates greater 5-HT1A potency than CBD suggests that decarboxylation does not uniformly improve receptor engagement across all targets. For this specific interaction, the carboxyl group appears to be a pharmacologically advantageous structural feature rather than an inert functional group awaiting removal.[18,22]
Third, 5-HT1A signaling intersects with nociceptive and inflammatory tone, providing a plausible mechanistic bridge between observed tolerability patterns and lipid signaling modulation discussed in adjacent subsections. This mechanistic framework also complements findings from the CBG sleep RCT, in which cannabigerol demonstrated clinically significant effects on sleep and quality-of-life outcomes.[23]
The preclinical data supporting this interaction constitute the strongest direct pharmacological evidence for the structural independence of CBDa from its neutral counterpart.[18,22] Whether 5-HT1A engagement at physiologically achievable plasma concentrations following oral dosing is sufficient to produce measurable clinical effects in humans remains to be confirmed. It is, however, a high-priority target for future clinical investigation.
How Does Arachidonic Acid Metabolism Create Cross-Talk Between Inflammatory and Endocannabinoid Signaling?
Arachidonic acid participates in lipid signaling networks that give rise to pro-inflammatory eicosanoids such as prostaglandins and leukotrienes and, through related biosynthetic pathways, to endocannabinoid signaling molecules, including anandamide and 2-AG.[19,24]
COX enzymes metabolize arachidonic acid and can also metabolize certain endocannabinoid derivatives.[24] This creates an intersection between inflammatory and endocannabinoid signaling networks. Inflammatory and endocannabinoid signaling do not operate as parallel systems. They share a biochemical lineage within broader lipid metabolism. Modulation of one lipid signaling pathway may influence tone in the other.[19]
This does not imply direct rebalancing of arachidonic acid metabolism by acidic cannabinoids. Rather, it highlights biochemical interconnections and reinforces that lipid signaling systems do not operate in isolation.[24,25]
How Does the Peripheral Distribution Model Apply to Nociceptive Signaling?
Pain signaling involves both central processing and peripheral nociceptive pathways.[26] Peripheral cannabinoid-responsive systems influence nociceptive thresholds.[20] Reduced central penetration combined with peripheral modulation may create a signaling profile distinct from intoxicating cannabinoids. For broader context on cannabinoid modulation of sleep-adjacent signaling, see .
The possibility of peripheral-biased modulation without central psychoactivity is consistent with currently understood structure and distribution logic. Whether this translates into meaningful clinical differences requires continued investigation. Distribution shapes interaction, and interaction contributes to outcome.[26,27]
What Does the In Vivo Conversion Question Imply for Peripheral Distribution Claims?
If acidic cannabinoids were universally and immediately converted to neutral forms upon ingestion, peripheral-biased modulation would be unlikely. However, acidic cannabinoids have been detected systemically after ingestion,[17] no rapid human decarboxylase has been identified,[13,17] and gastric acidity alone does not guarantee immediate conversion.[1,2]
The assumption of rapid in vivo decarboxylation has persisted for decades, yet direct mechanistic confirmation remains limited. As with other areas of pharmacology, assumptions can outlast the data supporting them.
Empirical real-world data from formulations containing stabilized acidic cannabinoids suggest that conversion is not total or instantaneous. Section V presents observational evidence consistent with incomplete in vivo decarboxylation and peripheral-biased distribution.
Does Modulation Require Intoxication to Be Biologically Relevant?
Pharmacology often equates perceptual magnitude with biological importance. That equation is convenient, but it is not necessarily correct.
Although acidic cannabinoids may not produce large perceptual shifts, their relevance, if present, may stem from tone modulation, enzyme interaction, peripheral signaling, and lipid pathway cross-talk.[18,19,21,24]
The absence of intoxication does not equate to the absence of biological interaction. Modern cannabinoid research has thoroughly characterized central CB1 agonism. The plant’s native acidic cannabinoid output remains comparatively underexplored. If acidic cannabinoids distribute and interact differently and are incompletely converted, then they cannot be reduced to precursors. They must be evaluated as primary molecular actors. Exploration requires restraint, precision, and stability control. It also requires separating assumption from evidence.
SECTION IV
Formulation Science and Molecular Fidelity: Why Preserving THCa and CBDa Integrity Is an Engineering Problem, Not a Sourcing Decision
If acidic cannabinoids represent the plant’s native cannabinoid chemistry, the next question is not whether they matter. It is whether they can be preserved. Working with acidic cannabinoids is not primarily a sourcing decision. It is a challenge of molecular stability and structural integrity. If structure determines interaction, then preserving structure becomes central to biological relevance.
Why Is Acidic Cannabinoid Formulation More Complex Than It Appears?
On paper, acidic cannabinoids seem straightforward: grow plant, extract, formulate. In practice, each step introduces risk of transformation. Acidic cannabinoids are chemically stable under specific conditions but vulnerable to environmental inputs that promote degradation. Exposure to heat, oxygen, light, moisture, mechanical disruption, and increased surface area all shift the equilibrium toward structural change.[1,28,29] Preserving acidic topology requires controlling variables that many conventional processing systems were never designed to manage.
Why Does the Crystalline State Offer Relative Stability, and What Conditions Destroy It?
Purified, crystalline THCa can remain relatively stable under controlled-temperature, low-moisture conditions.[data on file] However, stability is highly context-dependent. The risk of decarboxylation and oxidation increases when acidic cannabinoids are incorporated into high-surface-area matrices, suspended in oxygen-permeable oils, embedded in porous botanical material such as cured flower, exposed to repeated temperature fluctuations, or formulated in beverages or acidic liquids.[28,29]
Surface area accelerates exposure to oxygen. Oxygen accelerates oxidative transformation.[29] Time compounds both processes. The matrix in which acidic cannabinoids are formulated is not a secondary consideration. It is determinative.
This creates a formulation paradox. Large-surface-area technologies are often used to increase bioavailability and reduce onset time. Yet those same technologies increase oxidative exposure and structural instability. Therefore, preserving THCa integrity for oral or sublingual ingestion requires balancing competing priorities of absorption kinetics and molecular fidelity.[28]
Why Is Surface Area One of the Most Consequential and Underappreciated Variables in Acidic Cannabinoid Stability?
Surface area is one of the most consequential yet underappreciated variables in acidic cannabinoid stability. To understand why, it helps to distinguish between two physical states. A purified THCa crystal stored in a low-oxygen, low-moisture environment presents relatively little exposed molecular surface to its surroundings. The outermost layer of molecules interfaces with air, but the bulk of the crystal remains protected by its own lattice structure.
By contrast, when THCa is distributed across cannabis flower, milled into fine particulate matter, suspended in oil, or emulsified into a beverage, the exposed surface area increases dramatically. Instead of a compact crystalline structure, individual molecules or small aggregates are distributed across a much larger interface with oxygen, light, and moisture. A greater exposed surface area increases the probability of oxidative interaction.[28,29] Oxidation accelerates structural transformation, including decarboxylation and subsequent degradation.
For lipophilic compounds such as cannabinoids, increasing surface area is often desirable. A greater surface area can improve dissolution, enhance dispersion, increase absorption efficiency, and reduce onset time for oral or sublingual delivery. In other words, the same physical strategy that improves bioavailability also increases propensity for oxidation.[29]
In the flower, this tension is largely uncontrolled. Acidic cannabinoids are distributed throughout porous plant material with extensive surface exposure and environmental variability. In ingestibles and beverages, surface area is deliberately engineered to improve performance. Without protective formulation strategies, that increased exposure can accelerate the loss of acidic integrity over time.
The practical question that follows is whether the formulation delivered at the time of consumption reflects the structural composition implied at the time of testing. A certificate of analysis reflects a specific batch under specific laboratory conditions. It does not automatically guarantee structural integrity months later after exposure to oxygen, temperature fluctuation, light, and mechanical agitation during storage and distribution.[32,ICH] Therefore, preserving acidic topology extends beyond generating a compliant COA: it requires stability validation that accounts for real-world environmental stress.
The challenge is not simply preventing oxidation. It is balancing molecular accessibility with molecular preservation. Surface area improves interaction with human physiology, but it also increases interaction with the environment. Formulation decisions determine whether acidic cannabinoids maintain structural fidelity long enough to exert their intended effects.
What Strategies Exist for Reconciling the Bioavailability-Stability Trade-Off?
The tension between surface area and molecular stability is not irresolvable. Pharmaceutical science has developed several strategies for managing it, and their application to acidic cannabinoid formulations is an active area of technical development.[ICH]
Lipid-based delivery systems, including self-emulsifying drug delivery systems (SEDDS) and nanoemulsions, can improve dissolution and absorption of lipophilic compounds without requiring extreme particle size reduction.[Pouton] By dispersing cannabinoids within a structured lipid matrix rather than exposing them as free particulates, these systems can maintain bioavailability while reducing direct oxidative surface exposure. The lipid phase’s oxygen-barrier properties provide partial protection.
Controlled crystalline particle size represents a complementary approach. Rather than reducing particle size to the nanoscale and maximizing surface area, optimizing crystal dimensions to a range that balances dissolution rate with structural protection can reduce the surface area penalty. Pharmaceutical co-crystal engineering, in which the active molecule is co-crystallized with a stabilizing coformer, is an emerging strategy that can modify dissolution behavior without sacrificing the lattice-based protection of crystalline architecture.
Nitrogen-sparged encapsulation, in which headspace oxygen is displaced with an inert gas during filling and sealing, directly addresses oxidative exposure during manufacturing and storage. Oxygen-barrier packaging materials, opaque to relevant photochemical wavelengths, extend protection into distribution and retail environments.
None of these strategies eliminates the fundamental trade-off entirely. Each represents a partial solution with its own cost and complexity profile. The practical question for any acidic cannabinoid formulation is not whether the trade-off can be eliminated, but whether the specific combination of strategies employed is validated by real stability data across representative storage and distribution conditions rather than inferred from single-timepoint batch analysis.[ICH]
Why are Acidic Cannabinoids Especially Vulnerable to Heat During Processing?
Acidic cannabinoids are thermally labile and therefore temperature sensitive. Manufacturing processes that introduce heat, even moderate heat, can initiate partial decarboxylation.[1,2] Temperatures below combustion thresholds can still accelerate transformation over time.
Preserving acidic cannabinoids requires temperature disciplined control throughout extraction, post-extraction drying, post-processing, formulation, packaging, transport, and storage. Many existing cannabinoid supply chains were optimized for neutral cannabinoids and do not account for preservation of the plant’s primary biosynthetic chemical output as a primary constraint.[1,2,35]
How Does Photochemical Exposure Contribute to Cannabinoid Degradation?
Cannabinoids are photosensitive. Ultraviolet and certain visible wavelengths promote structural changes and degradation.[3] Opaque packaging and light-resistant storage are not merely aesthetic decisions. Rather, they are chemical safeguards.
Acidic cannabinoids exposed to sustained light degrade more rapidly than those shielded from photochemical energy input.[3] Stability is not passive. It must be engineered and continuously verified.
What Unique Challenges Do Aqueous and Beverage Formulations Present for Acidic Cannabinoid Stability?
Moisture introduces hydrolytic and oxidative risk. Acidic cannabinoids are not uniformly unstable in aqueous environments; however, prolonged exposure combined with oxygen and variable pH can accelerate transformation.[30]
Beverage formulations pose unique challenges stemming from dissolution dynamics, oxygen solubility, container permeability, and pH variability.[30] Stabilizing acidic cannabinoids in aqueous systems requires an advanced formulation strategy and rigorous stability validation. This complexity is often underestimated.
Dissolved oxygen is among the most consequential variables. Water can carry dissolved oxygen at concentrations sufficient to drive oxidative degradation of cannabinoids, particularly in the presence of light and elevated temperature.[29] Container headspace oxygen contributes an additional reservoir. Packaging materials with low oxygen transmission rates and nitrogen purging of headspace during filling are standard industrial mitigations, but their use in cannabinoid beverage formulations is inconsistent.
Water activity simultaneously influences microbial stability and chemical reactivity. At elevated water activity, molecular mobility increases, which can accelerate reaction kinetics, including oxidation and hydrolysis. pH variability adds a further complication. Acidic cannabinoids are carboxylic acids; their ionization state, and therefore their partition behavior and interaction with emulsification systems, shift with their pH. Formulations in low-pH environments, such as acidic beverages, alter the equilibrium between ionized and un-ionized forms, thereby affecting solubility, membrane permeability estimates, and emulsion stability.
Emulsification itself introduces surface area as a deliberate engineering choice for beverage applications. Nanoemulsified cannabinoid systems achieve rapid onset and good dispersion, but the resulting particle surfaces are exposed to the aqueous phase continuously. Without antioxidant co-formulation and appropriate emulsifier selection, oxidative degradation can proceed at commercially significant rates within weeks.[29,30] Stability claims for beverage-format acidic cannabinoids require validation across the expected shelf life and storage temperature range, not inference from day-of-production analytical results.[ICH]
Why Do Most Manufacturers Avoid Acidic Cannabinoid Formulations?
The relative scarcity of acidic cannabinoid formulations is not an artifact of botanical limitation. Acidic cannabinoids are abundant in the living plant. The constraint is structural preservation.
Working with acidic cannabinoids requires technical discipline throughout the entire supply chain. Temperature control during extraction and postprocessing becomes more consequential. Oxygen exposure during handling and encapsulation must be minimized. Packaging must account for light permeability and storage variability. Stability must be assessed over time rather than inferred from a single batch test.[ICH] Analytical verification must distinguish between acidic and neutral cannabinoid content without inducing transformation during measurement.[31]
Each of these considerations introduces operational complexity. Neutral cannabinoids are comparatively forgiving. Once decarboxylated, they tolerate environmental fluctuation with fewer structural consequences. Acidic cannabinoids do not offer that margin. They demand foresight, process alignment, and continuous validation.[1,28,29]
This difference in preservation requirements is not merely operational. It reflects how historical emphasis on transformed cannabinoids shaped production systems and expectations. When inquiry centers on transformation products, infrastructure evolves accordingly. Reorienting systems to preserve primary biosynthetic chemistry requires structural commitment.
The result is not that acidic cannabinoids are impractical. Rather, they require a different standard of process discipline. In a market optimized for throughput and tolerance of variability, structural preservation can seem inefficient. In a framework guided by molecular fidelity, structural preservation becomes necessary.
What Does Accurate Measurement of Acidic Cannabinoid Content Require?
If a product claims to contain acidic cannabinoids, the relevant question is not simply whether they were present at the time of testing. It is whether they remain present in the form and proportion implied by the label at the time of consumption.[ICH]
A certificate of analysis quantifies a batch under defined laboratory conditions. It does not inherently account for time, storage variability, distribution stress, or environmental exposure after testing. Acidic cannabinoids are sensitive to temperature, oxygen, light, and surface dynamics. Without stability data under realistic handling conditions, a single analytical snapshot may not fully represent the product’s ongoing structural integrity.[ICH]
Accurate quantification of acidic cannabinoids requires appropriate analytical methodology. High-performance liquid chromatography is necessary to differentiate acidic and neutral cannabinoids without inducing artificial decarboxylation during testing.[31] Gas chromatography, by contrast, can heat samples during analysis and convert acidic cannabinoids into their neutral forms, potentially misrepresenting composition.[31]
Equally important is whether the analysis differentiates between THCa and total THC. Reporting only total THC obscures the proportions of acidic and neutral content and does not clarify how much structural transformation may already have occurred. Transparency in reporting decarboxylation percentage, storage recommendations, and stability conditions reflects a commitment to molecular accuracy rather than regulatory minimalism.[31]
Precision in measurement is not a marketing detail. It is the foundation of structural fidelity. If the native chemistry thesis holds that topology influences distribution and interaction, then accurate, methodologically sound measurement becomes essential to preserving both scientific and consumer trust.
Why Does Preserving Molecular Topology Constitute Preserving Biological Function?
If acidic cannabinoids remain at least partially stable in vivo, as emerging evidence suggests,[17] then preserving their molecular topology during formulation becomes biologically relevant. If complete conversion occurred before systemic distribution, stability would be largely irrelevant. That question remains open.
Empirical real-world observations from formulations containing stabilized acidic cannabinoids suggest that conversion is not total or immediate. Preserving topology is therefore not an aesthetic preference. Rather, it is a mechanistic hypothesis grounded in structural logic. Section V presents observational data consistent with this inference.[Michigan pre-pub]
What Is the Practical Implication for the Industry?
Acidic cannabinoids are the primary biosynthetic metabolites of the cannabis and hemp plant. They are not scarce. They are difficult to preserve. Preservation requires chemical literacy, process discipline, stability testing, and transparent analytics.
Acidic cannabinoid formulation is a materials science challenge layered on top of pharmacology. That challenge explains why the field remains underdeveloped. It also explains why careful exploration is warranted.
SECTION V
Observational Evidence in Chronic Pain: Fibromyalgia, Rheumatoid Arthritis, and Osteoarthritis Across 162 Participants
The hypothesis that acidic cannabinoids may have underexplored biological relevance and exert meaningful biological effects independent of rapid in vivo decarboxylation and central intoxication required evaluation beyond mere structural reasoning. In collaboration with investigators affiliated with the University of Michigan, a 12-week prospective study allocated 162 participants across three cannabinoid formulation arms by three chronic pain populations: fibromyalgia (n=59), rheumatoid arthritis (n=26), and osteoarthritis of the knee and/or hip (n=76).[Michigan pre-pub]
Participants met criteria for substantial pain interference as measured by the PROMIS system and were assigned to one of three formulations: Arm 1, 12.5 mg CBD combined with 12.5 mg THC; Arm 2, 10 mg THCa combined with 10 mg CBDa, 5 mg CBG, and 3 mg CBC; and Arm 3, 10 mg CBD combined with 10 mg CBDa. A total of 162 participants completed the full assessment battery at baseline and the 12-week timepoint.[Michigan pre-pub]
What Was Observed Across Symptom Domains?
Across symptom domains, there was a statistically significant improvement over time.[Michigan pre-pub] Effect sizes, expressed as partial-eta squared, are presented in the table below.
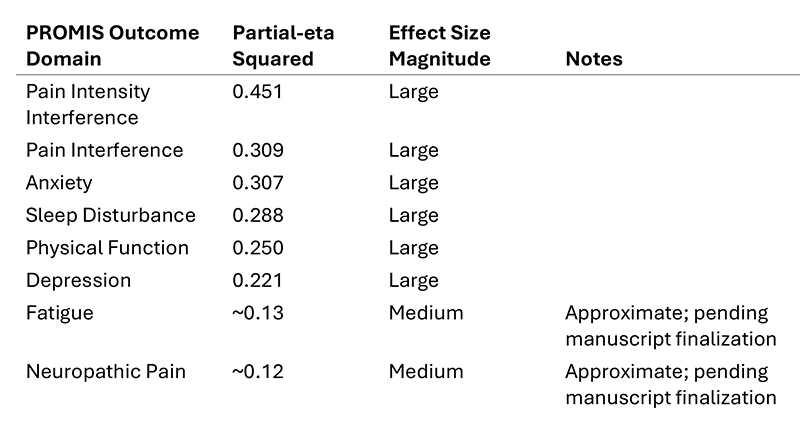
Importantly, improvements did not differ in magnitude across cannabinoid composition or chronic pain type.[Michigan pre-pub] This indicates that the formulation containing THCa, CBDa, CBG, and CBC performed comparably to the THC-containing formulation in overall symptom improvement.
The significance of this finding is structural rather than competitive. If acidic cannabinoids were universally and immediately converted to their neutral counterparts in vivo, one would expect outcomes to converge toward patterns dominated by central CB1-mediated effects. Instead, comparable improvements were observed without reliance on intoxicating doses of THC.[Michigan pre-pub]
Comparison of outcomes across formulation arms should be interpreted with appropriate caution. The study was not powered for between-group hypothesis testing, and no formal statistical correction for multiple comparisons across arms was applied. The observation that the acid-rich formulation arm did not demonstrate outcomes that were meaningfully lower in magnitude than the THC-containing arm represents an exploratory rather than confirmatory finding.[Michigan pre-pub]
What Does the Tolerability Profile Indicate About Mechanism?
An additional pattern emerged in the language participants used to describe their experience. Reports frequently referenced clear-headedness, absence of intoxication, functional stability, and reduced cognitive fog.[Michigan pre-pub]
These descriptions are consistent with reduced central CB1 engagement and peripheral-biased modulation, a structural prediction established in Section III. This does not establish a mechanism of action. It does reinforce coherence between structural pharmacology and lived experience.[6,13]
How Do Self-Reported Outcomes Relate to Plausible Enzyme Interaction Pathways?
Self-reported reductions in inflammatory discomfort align with plausible enzyme interaction pathways discussed in Section III, including COX modulation[21] and lipid signaling cross-talk.[19,24] Observational data cannot confirm enzyme inhibition in vivo. It can, however, demonstrate experiential patterns that are consistent with mechanistic plausibility. That is the appropriate role of real-world data: it lies between molecular hypothesis and controlled clinical validation.
Why Do These Findings Matter for the In Vivo Conversion Question?
If acidic cannabinoids were universally and immediately decarboxylated upon ingestion, distinctive experiential patterns would be unlikely. Clinical outcomes would be expected to resemble those of THC-dominant formulations alone.[Michigan pre-pub]
Instead, non-intoxicating cannabinoid compositions produced equivalent overall improvements in pain interference, sleep disturbance, and anxiety with large effect sizes across domains. This suggests that acidic cannabinoids may exert clinically relevant biological effects independent of central intoxication.[Michigan pre-pub]
More broadly, these observations return us to the central question of this document. If the plant consistently biosynthesizes cannabinoids in their acidic form, and if preserved acidic formulations can produce clinically meaningful outcomes without reliance on central intoxication, then the plant’s native chemistry cannot be dismissed as merely preparatory. It warrants evaluation as primary architecture.
The purpose of revisiting acidic cannabinoids is not to displace decades of research on neutral forms. It is to correct a structural imbalance in inquiry. Modern cannabinoid science has extensively characterized transformed chemistry. The observational data presented here suggest that the native biosynthetic state may deserve equal rigor.
What Are the Study Limitations, Including the BBB Caveat Specific to This Population?
A note on study limitations is essential. This observational study lacked a placebo arm, cell sizes were modest, and attrition among participants occurred. Although overall enrollment was substantial, stratification by pain condition and product arm resulted in smaller subgroup sizes, limiting statistical power for detecting differential effects.[Michigan pre-pub] The results, however, are consistent with the structural and pharmacokinetic reasoning outlined earlier in this guide and justify continued investigation under more controlled conditions.
One additional mechanistic consideration warrants explicit acknowledgment. The inference that acidic cannabinoids demonstrate reduced central nervous system penetration relative to THC rests on the general principle that increased polarity reduces passive diffusion across the blood-brain barrier under normal conditions, as established in Section III.[13] However, the study population, comprising individuals with chronic inflammatory pain conditions including fibromyalgia and rheumatoid arthritis, may have experienced degrees of blood-brain barrier disruption secondary to systemic inflammatory tone.
Neuroinflammation and peripheral inflammatory signaling can transiently increase BBB permeability,[32] potentially altering the distributional assumptions that underpin the peripheral-bias argument for acidic cannabinoids. This does not invalidate the structural reasoning, but it does mean that distributional predictions based on healthy-subject permeability models may not translate precisely to chronic inflammatory pain populations. Future pharmacokinetic studies in this population would benefit from explicitly measuring or controlling for this variable.
SECTION VI
Regulatory Status, Safety Boundaries, and the Limits of Current Evidence for Acidic Cannabinoid Formulations
Precision Requires Restraint
As interest in acidic cannabinoids grows, clarity becomes more important than enthusiasm. Acidic cannabinoids sit at an unusual intersection of plant biochemistry and emerging pharmacology. That intersection currently exists within regulatory ambiguity and the accelerating pace of commercial innovation.
If acidic cannabinoids are to be discussed seriously, boundaries must be defined as clearly as possibilities.
What Is the Current Federal Regulatory Status of THCa in the United States?
THCa occupies a particularly uncertain position in US federal regulatory interpretation. Unlike CBD, which received a specific scheduling exemption through the 2018 Farm Bill[33] for hemp-derived material meeting defined delta-9 THC thresholds, THCa has not received equivalent explicit federal clarification. The central ambiguity concerns whether THCa should be evaluated as a hemp-compliant compound based on its non-intoxicating native state or whether its potential to convert to delta-9 THC through decarboxylation places it within controlled substance scheduling calculations.
The DEA’s 2023 correspondence regarding hemp-derived psychoactive compounds,[DEA-2023] combined with ongoing state-level enforcement variability, creates a regulatory environment in which compliant product status for THCa-containing formulations is not uniformly determined. Some jurisdictions evaluate total THC potential, incorporating THCa via the decarboxylation conversion factor of 0.877 under [34]; others evaluate only the as-tested delta-9 THC content. This divergence has direct implications for labeling, distribution, and product classification.
Any formulation containing meaningful THCa concentrations must explicitly account for this regulatory variability. Compliance cannot be assumed from a single framework. Legal review specific to target markets, combined with transparent and methodology-specific analytical reporting, is non-negotiable. This is not a peripheral compliance consideration; it is a primary business and scientific integrity concern for this product category.
Does Non-Intoxicating Character Eliminate Regulatory Complexity?
THCa, CBDa, and other acidic cannabinoids are non-intoxicating in their native state. This is a structural reality grounded in reduced CB1 receptor engagement and altered distribution profiles. However, structural non-intoxication does not eliminate regulatory complexity.
Particularly in hemp-derived markets, THCa introduces specific considerations. Under certain conditions, including heat and environmental exposure, THCa can convert to THC through decarboxylation.[1,2] Therefore, storage, transport, and processing conditions matter. In many regulatory frameworks, total THC calculations incorporate potential decarboxylation.[34]
Clarity in labeling, stability testing, and reporting is non-negotiable. Responsible communication requires acknowledging both molecular identity and transformation potential.
How Does the Structural Conversion Question Connect to Legal Interpretation?
Earlier in this guide, we discussed the assumption that acidic cannabinoids rapidly decarboxylate in vivo. Although that hypothesis remains incompletely resolved, one principle is clear. Decarboxylation requires energy input or environmental transformation.
In the regulatory domain, that distinction has practical consequences. Acidic cannabinoids are chemically distinct from their neutral counterparts. Under certain conditions, they may convert. Precision in formulation must therefore be matched by precision in compliance.
What Do We Know About Safety, and Where Do the Gaps Remain?
Acidic cannabinoids share structural scaffolds with well-characterized cannabinoids. Under Threshold of Toxicological Concern frameworks, they do not introduce novel toxicophores outside established cannabinoid chemical classes.[14,15]
This provides a rational basis for continued investigation. It does not eliminate uncertainty. Long-term human data remain limited, as do large-scale randomized controlled trials. Dose-response relationships are not fully characterized, and vulnerable populations require cautious consideration.
Acidic cannabinoids should be approached in the same way as any biologically active compound. Dosing should begin conservatively. Consistency should be maintained to observe response patterns. Escalation without rationale should be avoided. Safety cannot be assumed but must be established through disciplined evidence.
For Which Contexts May Acid-Rich Formulations Be Appropriate, and for Which Are They Less So?
A recurring theme throughout this guide is modulation rather than blunt pharmacological force. Acidic cannabinoids may influence systems involving inflammatory signaling, nociception, stress-related tone, and physical discomfort.
Acidic cannabinoids do not cure autoimmune disease. They do not replace medical therapy or override behavioral dysfunction. They do not guarantee universal outcomes.
The observational study presented at ICRS 2024 demonstrated large improvements across chronic pain domains with multiple cannabinoid compositions, including acid-rich formulations.[Michigan pre-pub] Although the manuscript remains under peer review, the findings are consistent with the possibility that acidic cannabinoids may exert biologically relevant effects that are not reducible solely to rapid decarboxylation or central intoxication.
This does not establish a mechanism of action but bolsters the need for continued inquiry.
Acid-rich formulations may be appropriate when peripheral modulation is preferred, when central intoxication is undesirable, and when tolerance or avoidance of THC is relevant. They may be considered when inflammatory tone, nociceptive signaling, or physically driven sleep disruption contribute to overall symptom burden.
They may be less appropriate when acute severe pain requires rapid central analgesia, when severe sleep disorders require medical evaluation, or when behavioral misalignment dominates the underlying issue.
As with all cannabinoid use, clinical context determines appropriateness.
What Is the Risk of Over-Interpretation, and How Should the Scientific Position Be Stated?
The purpose of this guide has not been to argue that acidic cannabinoids are superior to neutral cannabinoids. It has been to examine whether the plant’s primary biosynthetic chemical output has received proportional scientific attention.
Modern cannabinoid science has extensively characterized chemically transformed cannabinoids. Revisiting the plant’s native state is not a rejection of prior work. It is an expansion of inquiry.
Evaluating acidic cannabinoids requires chemical understanding, stability control, transparent analytics, observational humility, and willingness to question assumptions. That includes the assumption that conversion renders the native molecule irrelevant.
The Closing Frame
Reinforcing the Thesis
Throughout this guide, we have examined acidic cannabinoids not as precursors to something more important but as the primary biosynthetic chemical output of the cannabis plant. Structural analysis (Sections I and II), stability considerations (Section IV), and observational data (Section V) suggest that their effects may not be reducible to rapid decarboxylation or central intoxication alone.
If that is correct, then evaluating acidic cannabinoids requires more than extracting and testing what remains after decarboxylation. It requires preserving topology, interrogating assumptions, and investigating biological interaction beyond perceptual magnitude.
The purpose of this document is not to elevate acidic cannabinoids above their neutral counterparts. It is to restore balance to the inquiry.
References
[1] Perrotin-Brunel H, Buijs W, van Spronsen J, et al. Decarboxylation of delta-9-tetrahydrocannabinol: kinetics and molecular modeling. J Mol Struct. 2011;987:67-73. https://doi.org/10.1016/j.molstruc.2010.11.061
[2] Wang M, Wang Y-H, Avula B, et al. Decarboxylation study of acidic cannabinoids: a novel analytical method with a theoretical explanation of heat-induced decarboxylation reactions. Cannabis Cannabinoid Res. 2016;1(1):262-271. https://doi.org/10.1089/can.2016.0020
[3] Casano S, et al. Photodegradation of cannabidiol (CBD) and delta-9-THC in cannabis plant material. Photochem Photobiol Sci. 2024. https://doi.org/10.1007/s43630-024-00589-4
[4] Taura F, Sirikantaramas S, Shoyama Y, et al. First cannabidiolic acid synthase from Cannabis sativa L. FEBS Lett. 2007;581(16):2929-2934. https://doi.org/10.1016/j.febslet.2007.05.043
[5] Sirikantaramas S, Taura F, Tanaka Y, et al. Tetrahydrocannabinolic acid synthase, the enzyme controlling marijuana psychoactivity, is secreted into the storage cavity of the glandular trichomes. Plant Cell Physiol. 2005;46(9):1578-1582. https://doi.org/10.1093/pcp/pci166
[6] Howlett AC, Barth F, Bonner TI, et al. International Union of Pharmacology. XXVII. Classification of Cannabinoid Receptors. Pharmacol Rev. 2002;54(2):161-202. https://doi.org/10.1124/pr.54.2.161
[7] Patrick GL. An Introduction to Medicinal Chemistry. 6th ed. Oxford: Oxford University Press; 2017. ISBN 978-0198749691.
[8] Long TW, Wagner M, Demske D, et al. Cannabis in Eurasia: origin of human use and Bronze Age trans-continental connections. Veget Hist Archaeobot. 2017;26:245-258. https://doi.org/10.1007/s00334-016-0579-6
[9] McPartland JM, Hegman W, Long T. Cannabis in Asia: its center of origin and early cultivation, based on a synthesis of subfossil pollen and archaeobotanical studies. Veget Hist Archaeobot. 2019;28:691-702. https://doi.org/10.1007/s00334-019-00731-8
[10] Swift W, Wong A, Li KM, Arnold JC, McGregor IS. Analysis of cannabis seizures in NSW, Australia: cannabis potency and cannabinoid profile. PLoS ONE. 2013;8(7):e70052. https://doi.org/10.1371/journal.pone.0070052
[11] Moreno T, Dyer P, Tallon S. Cannabinoid decarboxylation: a comparative kinetic study. Ind Eng Chem Res. 2020;59(46):20307-20315. https://doi.org/10.1021/acs.iecr.0c03791
[12] Huestis MA. Human cannabinoid pharmacokinetics. Chem Biodivers. 2007;4(8):1770-1804. https://doi.org/10.1002/cbdv.200790152
[13] Anderson LL, Low IK, Banister SD, McGregor IS, Arnold JC. Pharmacokinetics of phytocannabinoid acids and anticonvulsant effect of cannabidiolic acid in a mouse model of Dravet syndrome. J Nat Prod. 2019;82(11):3047-3055. https://doi.org/10.1021/acs.jnatprod.9b00600
[14] Munro IC, Ford RA, Kennepohl E, Sprenger JG. Correlation of structural class with no-observed-effect levels: a proposal for establishing a threshold of concern. Food Chem Toxicol. 1996;34(9):829-867. https://doi.org/10.1016/S0278-6915(96)00049-X
[15] Kroes R, Renwick AG, Cheeseman M, et al. Structure-based thresholds of toxicological concern (TTC): guidance for application to substances present at low levels in the diet. Food Chem Toxicol. 2004;42(1):65-83. https://doi.org/10.1016/j.fct.2003.08.006
[16] Mechoulam R. Plant cannabinoids: a neglected pharmacological treasure trove. Br J Pharmacol. 2005;146(7):913-915. https://doi.org/10.1038/sj.bjp.0706415
[17] Elder JH, et al. Pharmacokinetics and pharmacodynamics of a hemp-derived full-spectrum oral cannabinoid product. Cannabis Cannabinoid Res. 2025;10(2):e299-e313. https://doi.org/10.1089/can.2024.0187
[18] Bolognini D, et al. Cannabidiolic acid prevents vomiting in Suncus murinus and nausea in rats by enhancing 5-HT1A receptor activation. Br J Pharmacol. 2013;168(6):1456-1470. https://doi.org/10.1111/bph.12043
[19] Pacher P, Batkai S, Kunos G. The endocannabinoid system as an emerging target of pharmacotherapy. Pharmacol Rev. 2006;58(3):389-462. https://doi.org/10.1124/pr.58.3.2
[20] Pertwee RG, Howlett AC, Abood ME, et al. International Union of Basic and Clinical Pharmacology. LXXIX. Cannabinoid receptors and their ligands: beyond CB1 and CB2. Pharmacol Rev. 2010;62(4):588-631. https://doi.org/10.1124/pr.109.002501
[21] Takeda S, Misawa K, Yamamoto I, Watanabe K. Cannabidiolic acid as a selective cyclooxygenase-2 inhibitory component in cannabis. Drug Metab Dispos. 2008;36(9):1917-1921. https://doi.org/10.1124/dmd.108.020909
[22] Rock EM, Parker LA. Effect of low doses of cannabidiolic acid and ondansetron on LiCl-induced conditioned gaping in rats. Br J Pharmacol. 2013;169(3):685-692. https://doi.org/10.1111/bph.12162
[23] Emerson CR, Webster CE, Daza EJ, Klamer BG, Tummalacherla M. Effect of cannabigerol on sleep and quality of life in veterans. Med Cannabis Cannabinoids. 2026;9(1):1-14. https://doi.org/10.1159/000549902
[24] Rouzer CA, Marnett LJ. Endocannabinoid oxygenation by cyclooxygenases, lipoxygenases, and cytochromes P450. Chem Rev. 2011;111(10):5899-5921. https://doi.org/10.1021/cr2002799
[25] Hillard CJ. Circulating endocannabinoids: from whence do they come and where are they going? Int Rev Neurobiol. 2015;125:1-47. https://doi.org/10.1016/bs.irn.2015.10.001
[26] Hohmann AG, Suplita RL. Endocannabinoid mechanisms of pain modulation. AAPS J. 2006;8(4):E693-E708. https://doi.org/10.1208/aapsj080479
[27] Rock EM, Connolly C, Limebeer CL, Parker LA. Tetrahydrocannabinolic acid reduces nausea-induced conditioned gaping in rats and vomiting in Suncus murinus. Br J Pharmacol. 2013;170(3):641-648. https://doi.org/10.1111/bph.12316
[28] Bueno J, Alborzi S, Greenbaum EA. Vapor phase terpenes mitigate oxidative degradation of Cannabis sativa inflorescence. Cannabis Cannabinoid Res. 2023;8(5):887-898. https://doi.org/10.1089/can.2021.0207
[29] Singh D, et al. Antioxidants help favorably regulate the kinetics of lipid peroxidation, polyunsaturated fatty acids degradation and acidic cannabinoids decarboxylation in hempseed oil. Sci Rep. 2020;10:10563. https://doi.org/10.1038/s41598-020-67267-0
[30] Fraguas-Sanchez AI, Fernandez-Carballido A, Martin-Sabroso C, Torres-Suarez AI. Stability characteristics of cannabidiol for the design of pharmacological, biochemical and pharmaceutical studies. J Chromatogr B. 2020;1150:122188. https://doi.org/10.1016/j.jchromb.2020.122188
[31] Lazarjani MP, Young O, Kebede L, Seyfoddin A. Methods for quantification of cannabinoids: a narrative review. J Cannabis Res. 2020;2:35. https://doi.org/10.1186/s42238-020-00040-2
[32] Brooks TA, Hawkins BT, Huber JD, Egleton RD, Davis TP. Chronic inflammatory pain leads to increased blood-brain barrier permeability and tight junction protein alterations. Am J Physiol Heart Circ Physiol. 2005;289(2):H738-H743. https://doi.org/10.1152/ajpheart.01288.2004
[33] Agriculture Improvement Act of 2018, Pub. L. No. 115-334, 132 Stat. 4490.
[34] United States Department of Agriculture. Establishment of a Domestic Hemp Production Program. 7 C.F.R. Part 990 (2021). Decarboxylation conversion factor at section 990.1. Available at: https://www.ecfr.gov/current/title-7/subtitle-B/chapter-IX/part-990
[35] Fairbairn JW, Liebmann JA, Rowan MG. The stability of cannabis and its preparations on storage. J Pharm Pharmacol. 1976;28(1):1-7. https://doi.org/10.1111/j.2042-7158.1976.tb04014.x
Named-Key References
[Michigan pre-pub] Data on file, Metta Medical dba LEVEL. Kruger DJ, Dautrich T, Hall B, Keeling K, Provost K, Boehnke K. Assessing the efficacy of cannabinoid compositions for treating three classes of chronic pain. Presented at: International Cannabinoid Research Society Annual Symposium; 2024. University of Michigan IRB reviewed. Manuscript under peer review.
[data on file] Data on file, Metta Medical dba LEVEL. THCa crystalline stability under controlled temperature and low moisture conditions. Internal analytical data.
[ICH] International Council for Harmonisation. ICH Q1A(R2): Stability testing of new drug substances and products. Geneva: ICH; 2003. Available at: https://database.ich.org/sites/default/files/Q1A%28R2%29%20Guideline.pdf
[DEA-2023] Boos TL. DEA Letter: Re: Hemp and hemp-derived products, including delta-8-THC, delta-9-THCP, THC-O, HHC, THCA. Washington, DC: U.S. Drug Enforcement Administration; June 9, 2023. Available at: https://www.sog.unc.edu/sites/default/files/DEA-THCA-and-HHC-letter.pdf
[Pouton] Pouton CW. Formulation of poorly water-soluble drugs for oral administration: physicochemical and physiological issues and the lipid formulation classification system. Eur J Pharm Sci. 2006;29(3-4):278-287. https://doi.org/10.1016/j.ejps.2006.04.016

